
What Labs Need to Know About ISO 17025 Compliance in the Digital Era
When a pharmaceutical lab releases a new drug or a food testing facility clears products for consumption, lives literally depend on their accuracy. That’s why healthcare, food, environmental science, and construction remain among the most regulated industries.
Today, we’ll examine ISO 17025, one of the most critical standards that ensures laboratories in such industries produce traceable results.
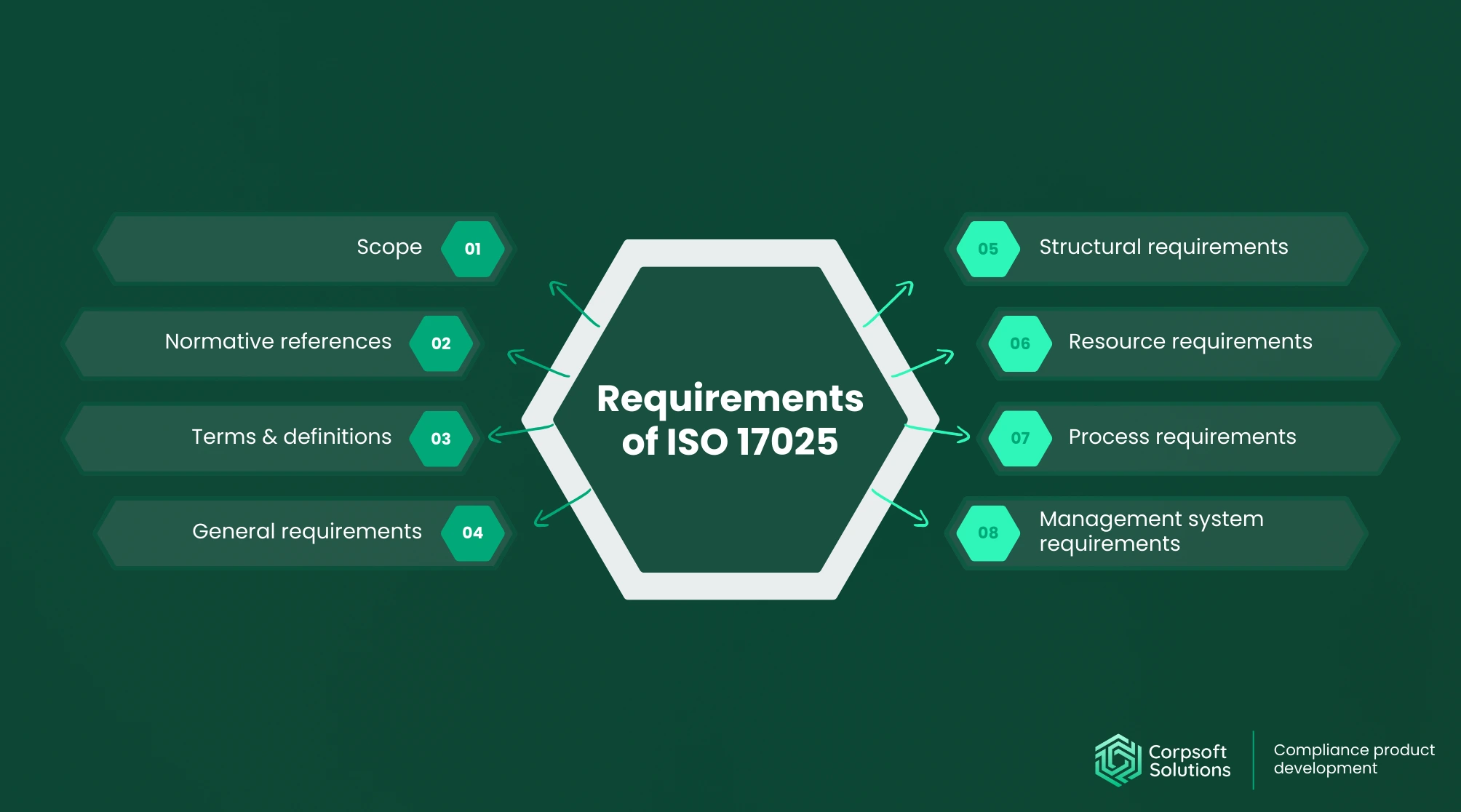
What ISO 17025 Certification Really Means
ISO 17025 is the international standard that defines the technical competence requirements for testing and calibration laboratories. Basically, it’s the guideline that sets the rules for how testing and calibration labs prove they know what they’re doing.
The primary goal of an ISO 17025 audit is to ensure that all measurement and testing results are accurate, reliable, and reproducible. This standard goes beyond the technical aspects of laboratory equipment or calibration. It helps you ensure that all your systems work in the best possible way.
In short, ISO calibration is not just about “let’s tune up all the scales,” but about creating a unified and reproducible system, including both software and hardware elements. It is suitable for both small private laboratories and large government or corporate centers.
But what sets ISO 17025 apart from other standards? Let’s make a recap:
- ISO 9001 focuses on overall quality management: internal processes, documentation, and customer satisfaction. But it doesn’t review the equipment accuracy.
- ISO 15189 is tailored to medical labs, addressing the specifics of clinical testing. Great for healthcare, but too narrow for other industries.
- ISO 27000 covers information security and data protection. They are still critical, but again, it’s not about lab measurements.
ISO 17025 emphasizes the technical competence of the laboratory itself, ensuring that every measurement and test is trustworthy. In other words, it combines quality management with concrete technical requirements, making it indispensable for any laboratory process.
Understanding what ISO 17025 means is just the first step. The real challenge lies in successfully navigating the audit process.
How To Pass ISO 17025 Audit
If you want to ensure that your testing facility will get certified, you must successfully pass an ISO 17025 audit. We highly recommend conducting your own mini-audit to identify improvement areas.
Digital tools can reduce audit preparation time by up to 60% and minimize human error in calibration tracking. However, implementing these tools without proper integration into existing workflows often creates more problems than solutions.
Let’s break down the ISO 17025 calibration requirements in more detail, so you know what to expect.
What Gets Checked During the Audit
An ISO 17025 audit focuses on whether your lab consistently delivers accurate and reliable results. Auditors don’t just look at the hardware, but also at how you manage it and whether you are strictly following internal procedures.
Here’s what gets checked during the ISP 17025 audit:
- Documentation and SOPs: up-to-date procedures, instructions, and policies that guide lab work.
- Equipment and calibration: instrument condition, calibration records, and maintenance logs.
- Staff and competence: qualifications, training, certifications, and staff assessments.
- Testing methods and validation: usage of recognized methods and traceability of measurement results.
- Handling of non-conformities and complaints: The way your lab records and resolves errors, and responds to client feedback.
- Digital systems and software: proper data management in LIMS, automated monitoring systems, and databases.
To pass the actual audit on the first try, you’ll need to prove that you are taking care of your laboratory by using the latest digital solutions and regularly calibrating all the equipment.
Basic ISO 17025 Checklist
Before applying for the actual audit, it’s worth checking ISO 17025 compliance requirements. To save you time, use our checklist with simple yet effective preparation tips.
| Item | How to Pass ✅ | How to Fail ❌ |
| Calibration and equipment verification records | Ensure that all instruments have up-to-date calibration logs; they should be dated and signed by the responsible person. | Miss the calibration deadline, forget about any records, or use instruments outside approved intervals. |
| SOP documentation (standard operating procedures) | Test and calibrate your equipment on time and ensure that these procedures are approved and easily accessible to staff. | Miss SOPs or ignore them for longer than the deadlines expect it. |
| Staff competence and training | Have up-to-date records of education, training, and certifications; ensure that your staff have passed required assessments. | You have no training records or staff who failed mandatory training. |
| Non-conformity and complaints management | You have established processes for recording errors, corrective actions, and root cause analysis. | There is no documentation, you refuse to track any errors, and you simply ignore client complaints. |
| Testing methods and traceability of results | You use proven methods, so it’s easy to track and repeat any lab experiment. | You don’t use standardized methods, so you can’t reproduce the same experiment. |
| Digital systems and software (LIMS, databases) | Your digital system records all results and calibration benchmarks. You regularly back up data and allow access only to authorized users. | You use unlicensed, unvalidated, or even pirated software. There are no backups, and your whole data is available even to unauthorised users. |
Regularly check the regulation updates, so you’ll know new ISO calibration requirements to fix gaps before the actual audit. It will increase your chances of passing on the first try.
Proper Calibration as the Foundation of ISO 17025 Compliance
Proper calibration is a fundamental element of this regulation. It helps you to ensure that all your measurements are accurate and reproducible. If you fail to meet calibration requirements, auditors may refuse to grant certification.
Luckily, you can automate this process with digital solutions. Just ensure that all your equipment is properly calibrated on time and that you have records of such tests.
An incorrect calibration interval of ISO 17025 can either lead to unnecessary expenses or the risk of using inaccurate results. So why spend extra money when you don’t have to?
Saving is the new earning.
ISO 17025 doesn’t have fixed calibration intervals, such as “check the microscope once a year”. Instead, each laboratory defines them based on its current workload.
| Factor | Impact on Interval | Example |
| Risk of equipment failure | The higher the impact of error on results, the shorter the interval. | Instruments used in pharmaceutical testing are calibrated more frequently than in a food lab. |
| Frequency of use | More frequent use → faster wear → shorter intervals | Scales that you use every day should be calibrated more often than a backup option. |
| Stability in past checks | If the equipment has been showing consistent results for several cycles, you can safely extend the interval. But not for too long, just made the interval 5-10% longer. | You need to calibrate the equipment less often than expected. If you need to test some scale once a year, you can extend the deadline for 1-2 weeks. |
| Customer or regulatory requirements | Some intervals may be mandated by law. | You need to obey the law. That’s all we can say. |
| Operating environment | Harsh conditions (humidity, dust, vibration) require shorter intervals | If you need to calibrate something every 6 months, but you use it in harsh conditions, test it every 4-5 months. Just in case. |
| Manufacturer recommendations | Suggested intervals serve as a starting point | If you’re still unsure about intervals, just look at the manual. If it states “Calibrate every 12 months,” do as described. |
Under ISO 17025 internal audit, calibration intervals are defined through a risk-based approach. They are not fixed dates written in stone, but decisions your lab makes based on real conditions.
Digital Tools for Tracking and Automating Calibration
![]()
It’s quite impossible to track all the calibrations in the modern laboratory manually. You may need to test dissectors every few days, scales every once in a week, microscopes every 2-4 months, so it’s easy to get lost.
No wonder that many laboratories that manually track it face the same question: “Am I really testing it, or am I just thinking about it for too long?”
That’s why advanced labs simplify this process by using the latest audit compliance software, such as LabWare LIMS, which helps you keep an eye on inventory, sends calendar reminders for calibrations, and stores all the data on the cloud.
Meanwhile, STARLIMS offers convenient lab management modules for calibration, SOP verification, and documentation. Qualtrax helps automate procedures, track calibration deadlines, and notify staff.
However, to ensure that this software gives you the best results, you need to make some hardware adjustments. Implement IoT sensors and digital loggers to monitor temperature, humidity, and instrument performance in real time.
But here’s the catch: you can’t just install a ready-made tool and expect it to magically work for your specific lab or clinic. Every solution needs to be adapted to your workflow. And that’s precisely where the Corpsoft Solutions team comes in.
Struggling to integrate tracking tools with your current laboratory workflow? Our team specializes in creating seamless digital solutions that align with ISO 17025 requirements from day one.
The Role of an ISO 17025 Consultant
Preparing for this certification will take a lot of time and effort. Many laboratories try to save money by doing everything themselves, but this carries a high risk of missing important details. And it may lead to a situation where the inspector may revoke your licence.
According to our experience, laboratories working with ISO 17025 consultants are 3x more likely to pass certification on the first attempt and reduce preparation time by an average of 4-6 months.
If you’re still asking yourself, “Do I need an ISO 17025 consultant to pass the certification?”, we wrapped it up in a simple table.
| Item | Self-Preparation | With an ISO 17025 consultant |
| Standard requirements analysis | It’s time-consuming because it’s easy to miss details. Since there is no one to help, the preparation may take months. | A consultant identifies critical points right away, shortening the preparation to weeks or even days. |
| Documentation & SOPs | Often incomplete, duplicated, or inconsistent. | All documents are structured, up-to-date, approved, and audit-ready. |
| Calibration & equipment tracking | Manual records pose a risk of incorrect intervals. | Integrated digital systems with proper intervals and full traceability. |
| Staff training | Training and competency checks are often inconsistent, which poses a high risk of non-compliance. | The consultant helps plan training and competency checks and prepares staff for the audit. |
| Risk of failing certification | There is a high risk since it’s easy to miss documentation gaps or minor errors during procedures. | The risk is low since all critical points are addressed, and the audit goes smoothly. |
| Time efficiency | Months of self-prep required. | Structured approach significantly reduces time to certification. |
We’ve already supported dozens of labs in passing ISO 17025 audits. One lab initially tried to manage everything alone but failed due to incorrect calibration intervals. So, they contacted us and asked the Corpsoft Solutions team to become their partners.
So, we designed and implemented a fully audit-ready digital system, automating reminders and internal tracking. With this solution, they have now passed three audits in a row, demonstrating the power of a tailored, tech-driven approach.
If your lab wants to simplify the workflow and meet ISO 17025 requirements, integrate digital monitoring tools. Corpsoft Solutions can help you with it, saving you time, reducing stress, and ensuring your lab is audit-ready.
Final Thoughts
ISO 17025 is much more than just a regulatory requirement. Today, compliance with this standard helps you to ensure your safety, strengthen a laboratory’s market position, and build client trust. Ready to make a change?
It’s time to embrace digital transformation with automated calibration, electronic documentation, and transparent processes. They will make your lab more efficient, modern, and competitive.
Want your lab fully audit-ready for ISO 17025? Corpsoft Solutions delivers digital tools that help you grow and become even more effective in this ever-changing world. From automated calibration to audit-ready documentation, we’ll help you develop and implement systems that meet all ISO 17025 technical requirements.
Subscribe to our blog


